The global marine ecosystem faces an estimated 11 million metric tons of plastic waste entering oceans annually (UNEP, 2025). Unlike conventional petroleum-based plastics that persist for centuries, Polyhydroxyalkanoates (PHAs) offer a technically viable, fully biodegradable alternative. PHAs achieve complete mineralization even in ambient marine environments, without requiring industrial composting—setting them apart from many “biodegradable” materials.
Microorganisms synthesize PHAs as natural energy reserves. Indigenous marine microbes readily metabolize their molecular structure through enzymatic hydrolysis, converting them into water-soluble monomers and ultimately CO₂, water, and biomass—with no persistent microplastics left behind.At SalesPlastics, our technical team sees growing demand for PHA resins in applications where end-of-life recovery is difficult, such as marine-sensitive packaging, aquaculture gear, and fishing nets.
This article explores PHA’s biological degradation mechanisms in marine settings, key influencing factors, and practical advantages—providing insights for brands transitioning to sustainable sourcing.
Curious if PHA suits your application?
Contact SalesPlastics for a free sample or customized performance comparison (PHA vs. PLA/PCL).
Physicochemical Properties and Marine Degradation Mechanisms
Polyhydroxyalkanoates (PHAs) are high-molecular-weight biopolyesters produced by microorganisms as intracellular storage compounds. Their structure consists of hydroxyalkanoate monomers linked by ester bonds, with poly(3-hydroxybutyrate) (PHB) being the most studied homopolymer, though PHA’s versatility comes from variable monomer compositions.
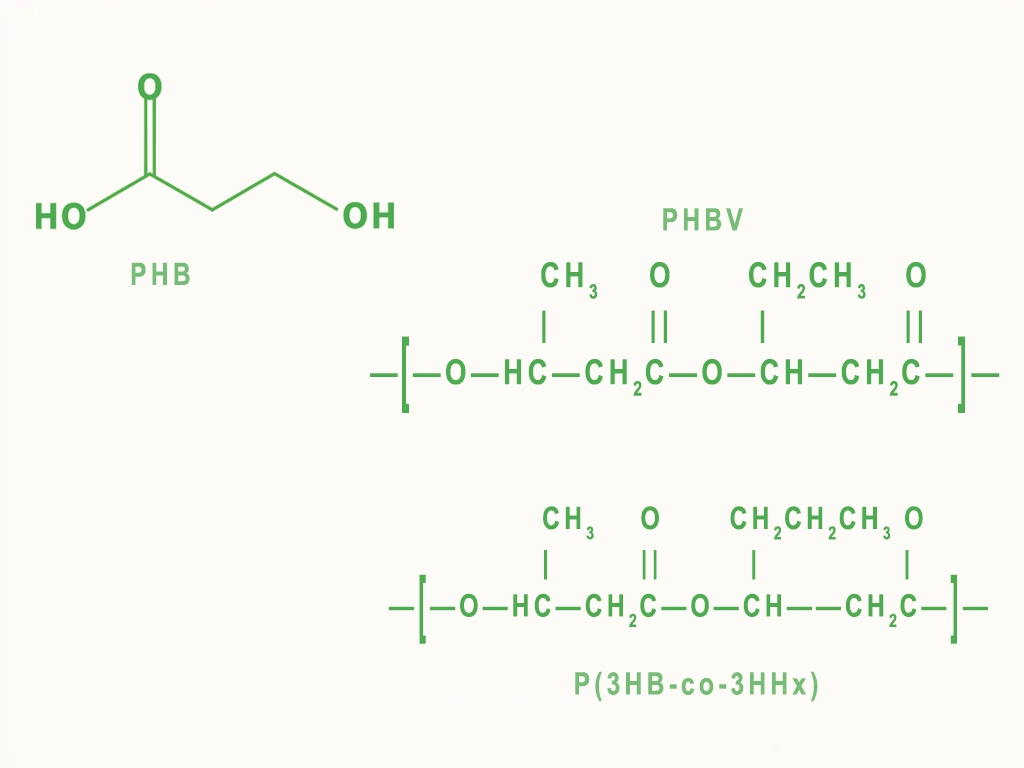
Chemical structures of common PHA types: PHB, PHBV, and PHBHHx
Properties vary with side-chain length: short-chain-length PHAs (scl-PHAs) like PHB offer high crystallinity and strength similar to polypropylene, while medium-chain-length PHAs (mcl-PHAs) provide elastomeric flexibility. At SalesPlastics, we leverage this tunability to supply grades tailored to specific mechanical and barrier needs.
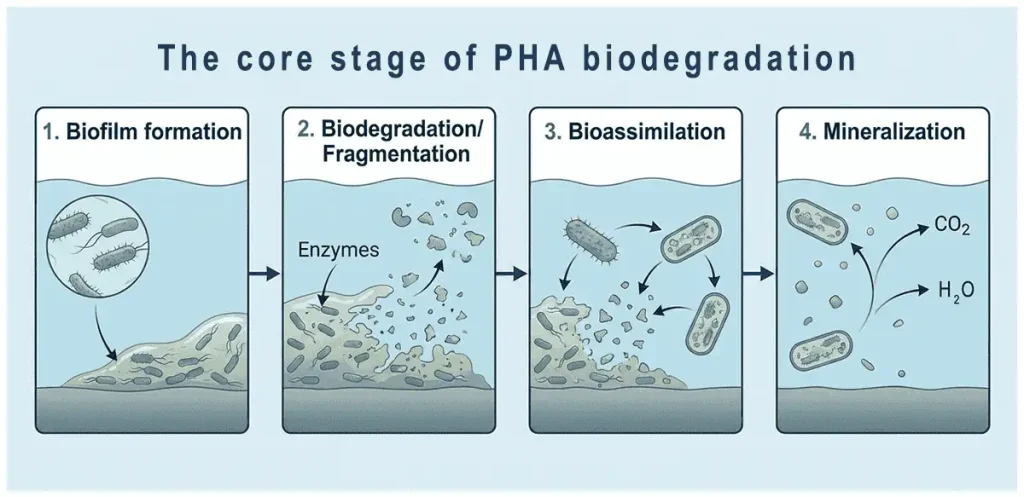
Sequential stages of PHA biodegradation in marine environments
PHA’s marine superiority stems from a four-stage enzymatic process:
- Biofilm Formation: Marine microbes colonize the surface, secreting enzymes.
- Bio-fragmentation: Extracellular depolymerases cleave ester bonds into oligomers and monomers.
- Bio-assimilation: Monomers enter microbial metabolism (e.g., β-oxidation, TCA cycle).
- Mineralization: Final products are CO₂, water, and biomass (methane possible anaerobically).
PHA degrades via surface erosion, maintaining integrity longer than bulk-degrading polymers, ensuring functionality before full integration.
Core Advantages of PHA in Marine Degradation
PHAs achieve total mineralization without residues, unlike fossil plastics that fragment into microplastics. Marine life interacts safely with degrading PHA.

Marine organisms can safely interact with degrading PHA materials without harmful effects
However, while PHA excels environmentally, challenges like higher costs (currently 2-5 times traditional plastics) and processing sensitivity exist. These are improving with scale-up.
Comparison with Non-Biodegradable Plastics and Other Bioplastics
To understand why PHA is the leading marine degradable bioplastic, it is essential to compare its environmental performance against industry benchmarks like PE and PLA.”
| Material Type | Marine Biodegradability | Est. Degradation Time | Ecological Impact |
|---|---|---|---|
| Conventional (PE/PP) | Negligible | Centuries | High; persistent accumulation |
| PLA | Minimal | >3–5 Years | Persistent in cold seawater |
| PHA | Complete | 3–18 Months (films); 1.5–4 Years (thicker items) | Minimal; becomes nutrient source |
| PCL | Moderate | 12–36 Months | Low; slower in cold conditions |
PHA succeeds in seawater due to temperature independence, metabolic compatibility with marine microbes, and absence of toxic additives.For recoverable products, PLA may be cost-effective; for high-risk marine leakage (e.g., fishing gear), PHA is ideal.
Factors Affecting Marine Degradation of PHA
The degradation kinetics of PHA in marine settings are not uniform; they are governed by a complex interplay between extrinsic environmental variables and intrinsic material properties. For technical teams and product developers, understanding these factors is essential for predicting the service life and environmental fate of biopolymer applications.
Environmental Factors
- Temperature (faster in tropical waters via Q10 effect)
- Oxygen levels
- UV synergy
- Microbial density (higher in coastal areas).
Material Factors
- Copolymerization reduces crystallinity for faster degradation;
- Surface-to-volume ratio critical (thinner items degrade quicker);
- Molecular weight and processing affect onset.
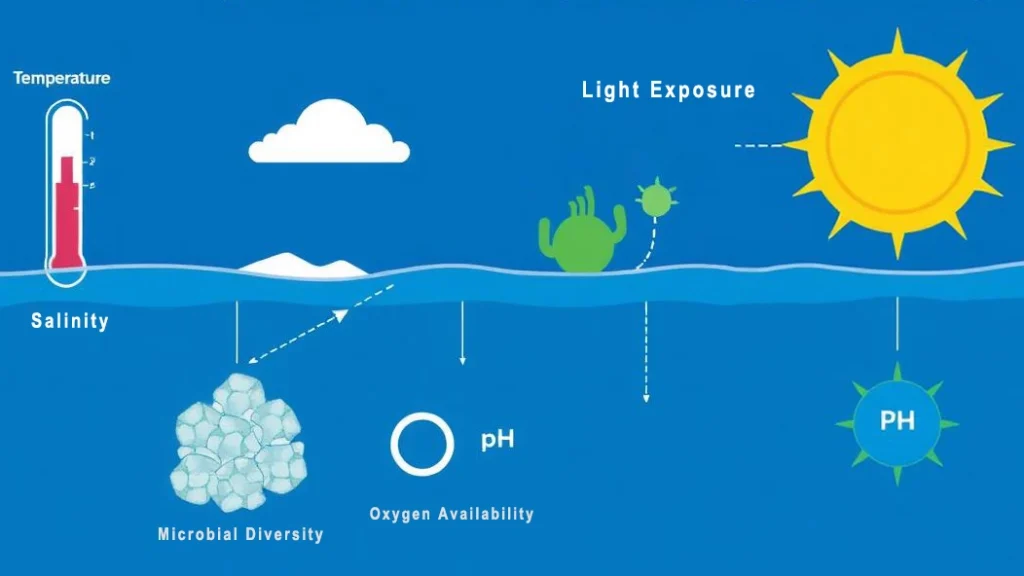
Key environmental factors influencing PHA biodegradation rates in marine settings
Need a custom degradation forecast?
SalesPlastics offers performance assessments for your environment.
Degradation Timeframes in Different Marine Conditions
The combined effect of environmental and material factors results in a wide range of degradation timeframes for PHA materials in different marine conditions. Understanding these timeframes is essential for designing appropriate applications and setting realistic expectations for PHA performance in marine environments.
| Marine Environment | PHA Type | Material Form | Approximate Degradation Time | Key Factors |
|---|---|---|---|---|
| Tropical surface (25-30°C) | PHBV (12% HV) | Thin film (100 μm) | 1-4 months | High temp, UV, microbes |
| Temperate coastal (15-20°C) | PHB | Thin film (100 μm) | 4-12 months | Moderate temp, diversity |
| Temperate coastal (15-20°C) | PHB | Bottle (1-2 mm) | 1.5-4 years | Thickness, crystallinity |
| Cold waters (5-10°C) | PHBV | Thin film (100 μm) | 8-18 months | Low temp, reduced activity |
| Deep sea sediments | PHB | Pellets (3-5 mm) | 3-6 years | Low oxygen, temp |
| Estuarine | P(3HB-co-3HHx) | Thin film (100 μm) | 2-6 months | High nutrients, microbes |
Mean rate: ~0.04-0.09 mg·day⁻¹·cm⁻². Pattern often includes initial lag, rapid phase, then slower mineralization. Actual times vary.
Application Significance and Prospects of PHA
The structural versatility of Polyhydroxyalkanoates (PHAs) enables their deployment in environments where conventional polymers pose the highest ecological risks. By aligning material performance with environmental safety, PHAs address the critical “end-of-life” gap in marine-sensitive industries.
Targeted Marine Applications
Current adoption focuses on sectors where material recovery is statistically improbable or logistically complex:

Marine applications of PHA materials: fishing gear, aquaculture components, and marine construction
Aquaculture/fishing
Development of high-tenacity nets, longlines, and oyster mesh that mitigate “ghost fishing” risks. If lost, these components undergo enzymatic erosion, preventing decades of entanglement.
Marine engineering
Biodegradable sensors, benthic monitoring casings, and temporary construction barriers. These devices fulfill their functional lifecycle before integrating into the sediment.
Coastal packaging
Single-use items for cruise lines and beach venues—such as cutlery, straws, and coatings—where leakage into the marine environment is a persistent challenge despite waste management efforts.
Market projected to grow at 16.5% CAGR (2025-2030), driven by regulations and ESG goals. Hurdles include costs (mitigated by waste feedstocks and scale) and processing (improved via blending/additives).
Future Research Directions
The next frontier for PHA involves multi-disciplinary research to bridge the gap between laboratory performance and real-world durability:

Research on PHA marine biodegradation combines materials science, microbiology, and marine ecology
Precision Degradation
Developing “intelligent” copolymers where degradation onset is triggered by specific environmental markers, ensuring durability during service and rapid breakdown thereafter.
Ecological Impact Validation
Longitudinal field studies are essential to confirm that PHA mineralization products—beyond being non-toxic—do not disrupt localized microbial community structures.
Regulatory Standardization
Establishing globally recognized marine biodegradation protocols is paramount. Moving beyond laboratory-simulated seawater to standardized field-testing will provide the certainty required for large-scale industrial adoption.
Conclusion
Polyhydroxyalkanoates (PHAs) offer a technically validated framework for addressing the persistent challenge of marine plastic pollution. By aligning material functionality with authentic environmental integration, PHAs serve as a critical bridge for applications where mechanical performance is required but material recovery remains logistically unfeasible.
The core advantages of PHA in marine settings are summarized as follows:
- Total Mineralization: Complete enzymatic conversion into CO₂ water, and biomass, effectively eliminating the risk of microplastic accumulation.
- Innate Biocompatibility: Seamless integration with existing marine microbial pathways without requiring high-temperature industrial triggers.
- Tunable Degradation Profiles: Predictable degradation kinetics that can be engineered through molecular architecture and surface modifications.
- Ecological Safety: A benign lifecycle that avoids the leaching of toxic plasticizers or endocrine-disrupting additives into aquatic habitats.
Despite ongoing economic/processing challenges, expanding production and regulations support adoption. SalesPlastics partners with manufacturers for seamless PHA integration – from resin selection to application testing.
Ready to transition?
Contact our experts for quotes, samples, or marine-grade PHA solutions.
FAQs
Is PHA marine degradable?
Yes, PHAs are fully marine-degradable. Unlike many bioplastics that require industrial composting conditions (high temperature and specific humidity) to fragment, PHAs undergo natural mineralization in aquatic environments. This process is driven by indigenous marine microorganisms that secrete extracellular enzymes known as PHA depolymerases. These enzymes hydrolyze the polymer chains into water-soluble monomers, which are subsequently metabolized by the microbiota. Extensive research confirms that PHAs leave no persistent microplastics or ecotoxic residues across various settings, from coastal waters to deep-sea sediments.
What is the degradation time of PHA?
The degradation timeframe depends on the specific polymer grade, material geometry, and localized environmental variables. Technical data indicates a mean biodegradation rate of approximately 0.04-0.09 mg·day⁻¹·cm⁻² in marine settings. Under these parameters, a thin PHA film (100 μm) typically mineralizes within 1–6 months. In contrast, thicker rigid structures, such as bottles (1-2 mm), may require 1.5–3.5 years for complete biological integration. Performance varies geographically, with accelerated kinetics in nutrient-rich tropical waters compared to colder, deep-ocean environments.
How long does it take for PHA to break down?
The breakdown process is a phased biological progression: initial microbial colonization is followed by surface erosion and eventual metabolic assimilation. While a 100 μm film often breaks down within 6 months, medium-gauge items (0.5-1 mm) generally integrate into the environment within 12 months. This timeline is heavily influenced by the Q10 temperature coefficient; for every 10℃ increase in seawater temperature, microbial metabolic rates approximately double. Consequently, PHA products will transition to CO₂, water, and biomass twice as fast in tropical climates (25-30℃) as they do in temperate zones (5-10℃).
What are the problems with PHA?
While PHAs offer significant environmental benefits, several industrial challenges are currently the focus of ongoing research and development:
- Production Economics: Current costs remain 2–5 times higher than conventional polyolefins due to complex fermentation cycles and nascent economies of scale.
- Mechanical Brittleness: Certain grades, particularly PHB homopolymers, exhibit high crystallinity and a narrow processing window, though this is mitigated through copolymerization.
- Processing Sensitivity: PHAs often require precise thermal management and specific screw designs to prevent molecular weight reduction during extrusion or injection molding.
- Batch Consistency: Maintaining identical physical properties across large-scale microbial production runs remains a technical focus for resin manufacturers.
- Storage Requirements: The resin’s inherent biodegradability necessitates controlled storage conditions to prevent premature degradation in environments with extreme humidity or temperature.



